Acid and Base: Acid Vs Base
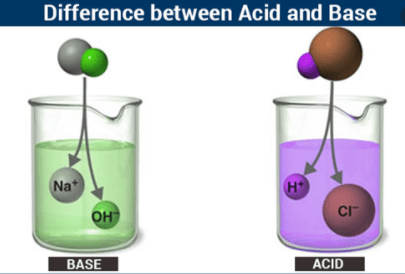

The Basic Difference between acid and Base is that Acids are substances with a high concentration of hydrogen ions and whose pH is lower than 7. A base is an ion or molecule that can provide protons or capture electrons, it is designated by the ion HO- and when an acid is neutralized with it, can lead to salts. The pH of a base is greater than 7.
Acid
It is a substance that, in solution, can increase the concentration of hydrogen ions. There are a large number of acids and one of the most elaborated in the industry is sulfuric acid, of the chemical formula H2SO4 is very corrosive and reacts violently with water.
Acid is also defined as a substance capable of emitting a proton or accepting electrons to achieve stability.
They have a pH lower than 7, they are abrasive, they release hydrogen when mixed with active metals, they are vicious, they can burn the skin, they destroy biological tissues, when they come into contact with marble they create effervescences and their properties disappear when they interact with salts.
Base
A base is defined as an ion or molecule that has the ability to provide electrons or capture protons. It is described with the hydroxyl ion HO-. When used to neutralize an acid, it will form a salt.
Among the best-known bases are caustic soda or sodium hydroxide, potash or potassium hydroxide, and ammonia.
Bases have a pH greater than 7 and are as corrosive as an acid.
Differences between acid and base
- An acid is a substance that has a high concentration of hydrogen ions.
- Acids have a pH of less than 7.
- Acids release hydrogen ions when they mix with water.
- A base is a molecule or ion capable of taking up protons or providing electrons.
- A base joining with an acid forms salts.
- Bases have a pH greater than 7.